Clinical
trials
at the MC "MEDHOUSE"
The medical center is accredited by the
MoH of Ukraine.
Ivano-Frankivsk,
22a Mykhaila Hrushevskoho Str.

More than
10 years of experience
The MC "MEDHOUSE" employs highly qualified doctors with long-term experience in conducting international randomized Clinical Trials.
Our study team members have valid GCP (Good Clinical Practice) certificates, as well as IATA certification for dangerous goods packaging and shipping according to 49CFR 172.700 / IATA 1.5
Clinical Trials at the Medical Center "MEDHOUSE" are conducted in accordance with the current requirements of Ukrainian legislation and international recommendations.
The Local Ethics Committee was formed and is actively functioning at our Center.
If you are interested in cooperation in the field of Clinical Trials, please contact us
Areas of clinical trial conduct at the MC "MEDHOUSE"
Best conditions for Clinical Trials conduct and monitoring according to GCP
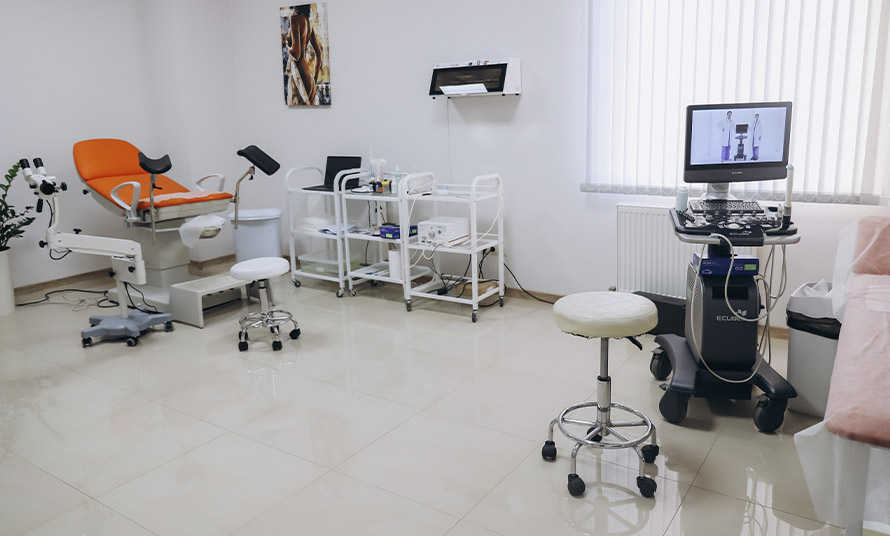
Equipment
All Clinical Trials related equipment is appropriately metrologically certified. There are all necessary conditions for study drug, documentation and laboratory samples storage
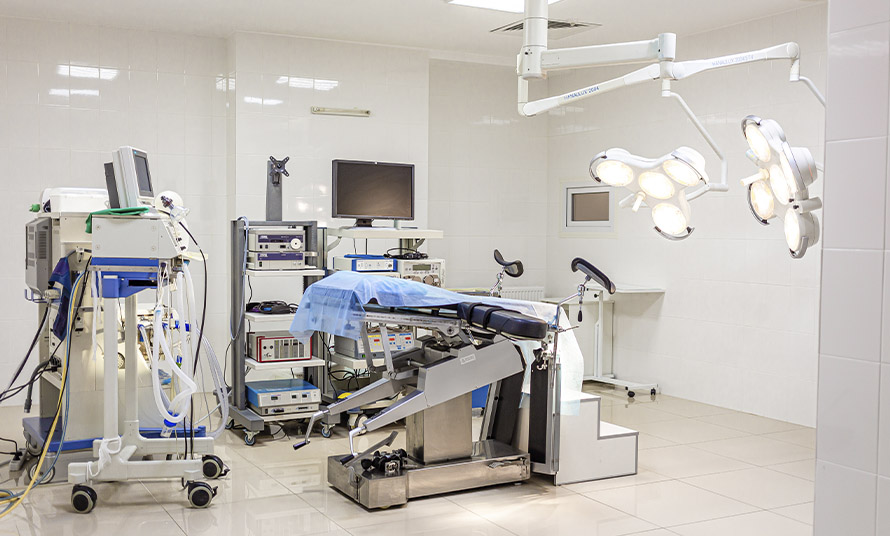
Operating room
Modern operating room

Intensive care unit
There is an intensive care unit with the possibility of providing emergency medical care

Inpatient department
Modern wards for inpatients

Day hospital
Comfortable rooms for intravenous administration of drugs, including chemotherapy
Best conditions for Clinical Trials conduct and monitoring according to GCP
We cooperate with
Carpathian Research Group -
Global Contract Research & Site Management Organization (CRO) conducting and supporting Clinical Trials of pharmaceuticals and medical devices.

Become a partner of the MC "MEDHOUSE"
For cooperation in the field of Clinical Trials for Contract Research Organizations, please contact the project coordinator at the MC "MEDHOUSE".
Volodymyr Galyuk
MD, PhD, Associate Professor

Contact
Contact us to get more information about Clinical Trials
Address
Ivano-Frankivsk,
22a Mykhaila Hrushevskoho Str.


 English
English  Українська
Українська 



